- Blog
- Roblox jailbreak free money vip server
- Stick ranger 2 download
- Print your brackets super bowl squares
- Macbook photo booth online
- Sinhala novels sujeewa prasanna arachchi
- Nba 2k14 generic mod enabler ubr
- Wedding planner budget template
- Fate hollow ataraxia caren is 14
- Avast free crack serial
- Unity funeral home harlem nyc
- Online soccer tactic board
- Ff14 reshade preset
- Online color picker upload image
- Rome 2 total war blood and gore
- Internal audit checklist template iso 13485
- Combat flight sim 1 no cd crack
- Sony vegas pro 13 trial serial number
- Pro valorant players crosshair
- Ck2 dlc unlocker mac
- World war ship with airplane coloring page
- Best free app to edit youtube videos
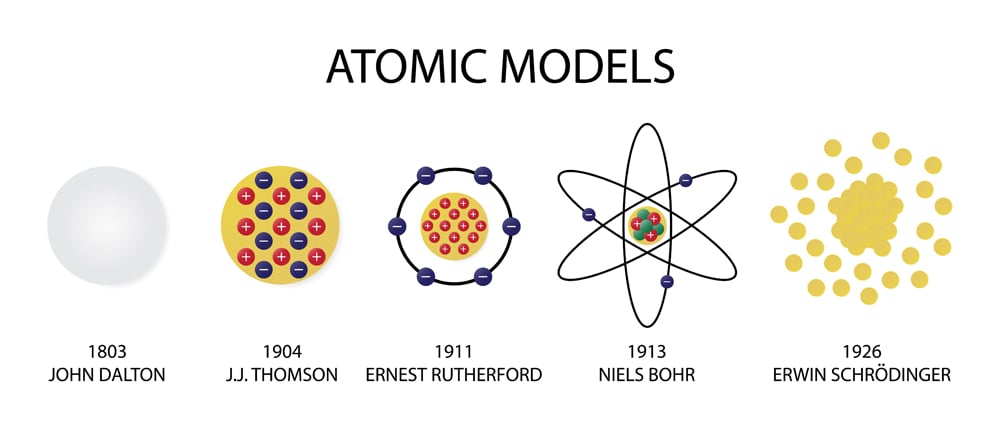
- The atomic theory
- Gta 5 free download on pc
- Diabetic meal plan chart for large male
- Expense tracker excel spreadsheet
- Fortnite for the damaged coda
- Macos mojave - dmg
- Unlocked games bloons tower defense 5
- Color finale pro 1-8-2
- Download bejeweled 2 full version free
- Xforce keygen autocad 2012 64 bit --
- Hans decoz numerology free pdf
- Free full movie percy jackson lightning thief
- Travel expense report template excel
- Charles daughter charles barkley daughter
- Old windows xp file shredder
- Density of water formula
- Birthday sex jeremih song lyrics
- Joy pony game free no download
- Bts merch sims 4 cc
- Gta 6 leak video reddit
- Super health club walkthrough watch movie
- App to erase background on pictures
A very rare form of hydrogen has one proton and two neutrons in the nucleus this isotope of hydrogen is called tritium. This particular isotope of hydrogen is called deuterium. However, a small number (about one in a million) of hydrogen atoms have a proton and a neutron in their nuclei. For example, most hydrogen atoms have a single proton in their nucleus. Most naturally occurring elements exist as isotopes. Atoms of the same element (i.e., atoms with the same number of protons) with different numbers of neutrons are called isotopes. Each element has its own characteristic atomic number.Ītoms of the same element can have different numbers of neutrons, however. Thus, hydrogen has an atomic number of 1, while iron has an atomic number of 26. This number of protons is so important to the identity of an atom that it is called the atomic number of the element. All atoms of hydrogen have one and only one proton in the nucleus all atoms of iron have 26 protons in the nucleus. What makes atoms of different elements different? The fundamental characteristic that all atoms of the same element share is the number of protons. The modern atomic theory states that atoms of one element are the same, while atoms of different elements are different. (See Figure 3.4 “The Structure of the Atom”.) Figure 3.4 “The Structure of the Atom.” Atoms have protons and neutrons in the centre, making the nucleus, while the electrons orbit the nucleus. The electrons are outside the nucleus and spend their time orbiting in space about the nucleus. The relatively massive protons and neutrons are collected in the centre of an atom, in a region called the nucleus of the atom (plural nuclei).
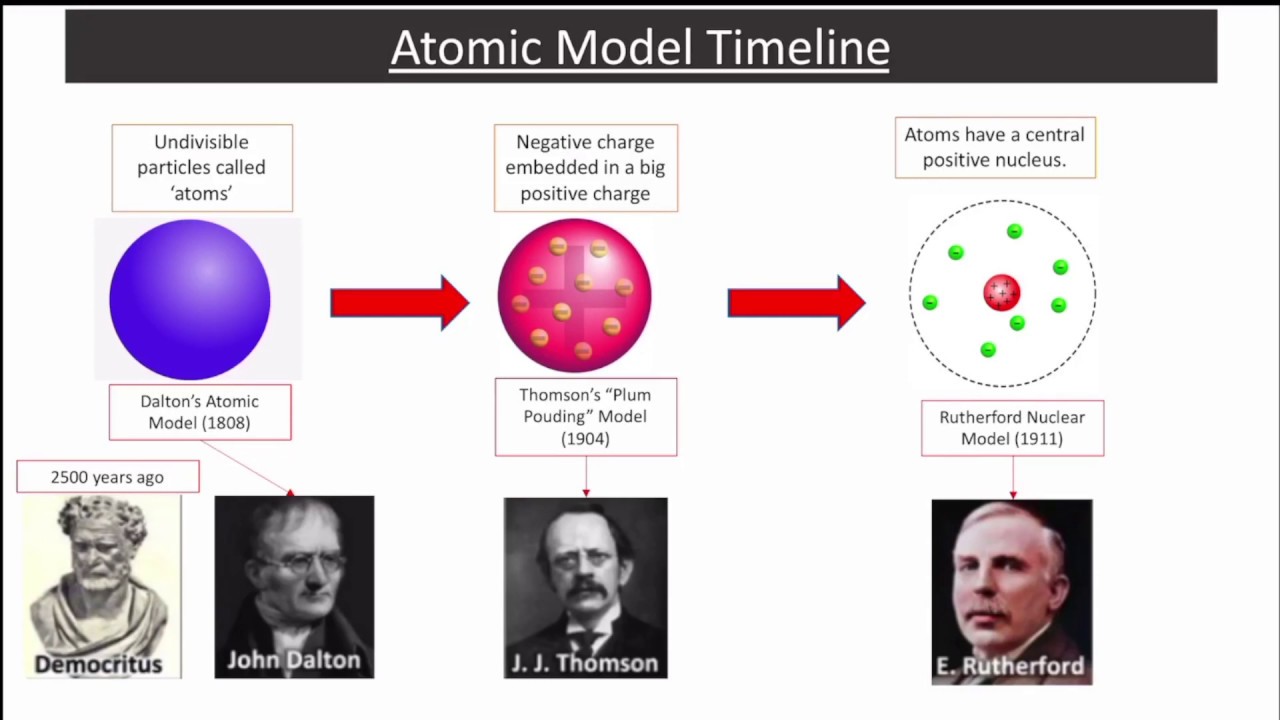
Experiments by Ernest Rutherford in England in the 1910s pointed to a nuclear model of the atom. How are these particles arranged in atoms? They are not arranged at random. Table 3.7 Properties of the Three Subatomic Particles Name

Table 3.7 “Properties of the Three Subatomic Particles” summarizes the properties of these three subatomic particles. We now know that all atoms of all elements are composed of electrons, protons, and (with one exception) neutrons. The neutron is a subatomic particle with about the same mass as a proton but no charge. The proton is a more massive (but still tiny) subatomic particle with a positive charge, represented as p +. Later, two larger particles were discovered. It is often represented as e −, with the right superscript showing the negative charge.

The first part to be discovered was the electron, a tiny subatomic particle with a negative charge. These concepts form the basis of chemistry.Īlthough the word atom comes from a Greek word that means “indivisible,” we understand now that atoms themselves are composed of smaller parts called subatomic particles. Atoms combine in whole-number ratios to form compounds.Atoms of the same element are the same atoms of different elements are different.The concept that atoms play a fundamental role in chemistry is formalized by the modern atomic theory, first stated by John Dalton, an English scientist, in 1808. Atoms are so small that it is difficult to believe that all matter is made from atoms-but it is. The period at the end of a printed sentence has several million atoms in it. It would take about fifty million atoms in a row to make a line that is 1 cm long. The smallest piece of an element that maintains the identity of that element is called an atom.
